Using the data in the table, which of the conjugate acids below is the strongest acid? 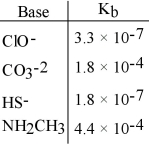
A) HClO
B) HCO3-
C) H2S
D) NH3CH3+
E) H2S and HClO
Correct Answer:
Verified
Q10: Classify the following compounds as weak acids
Q20: Which solution below has the highest concentration
Q21: A- is a weak base. Which equilibrium
Q22: HA is a weak acid. Which equilibrium
Q28: Using the data in the table, which
Q29: In which of the following aqueous solutions
Q30: Using the data in the table, which
Q33: Classify the following compounds as weak bases
Q35: Ammonia is a _.
A)weak acid
B)strong base
C)weak base
D)strong
Q36: Classify the following compounds as weak acids
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents