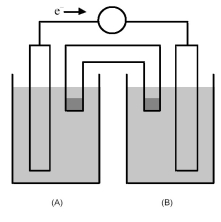
-The cell reaction 2 Fe3+(aq) + Zn(s) → Zn2+(aq) + 2 Fe2+(aq) occurs in the galvanic cell shown above.Which would be the most appropriate choices for the solid electrode in half-cell (A) and in half-cell (B) ?
A) Fe(s) for half-cell (A) and Zn(s) for half-cell (B)
B) Pt(s) for half-cell (A) and Zn(s) for half-cell (B)
C) Fe(s) for half-cell (A) and Fe(s) for half-cell (B)
D) Zn(s) for half-cell (A) and Pt(s) for half-cell (B)
Correct Answer:
Verified
Q115: The initial concentrations of Ag+(aq)and Cu2+(aq)are both
Q116: Fuel cells
A)produce carbon dioxide and hydrogen.
B)emit sulfur
Q117: Consider the galvanic cell shown below.
Q118: Q119: What is the shorthand notation for this Q121: Based on the balanced chemical equation shown Q122: According to the balanced equation shown below,3.00 Q123: The initial concentrations of Ag+(aq)and Cu2+(aq)are both Q124: Consider the following galvanic cell. Q125: Shown below is an electrochemical cell with![]()

Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents