In the Haber process,ammonia gas is synthesized from nitrogen gas and hydrogen gas.ΔG° at 298 K for this reaction is -33.3 kJ/mol.The value of ΔG at 298 K for a reaction mixture that consists of 1.9 atm nitrogen gas,2.3 atm hydrogen gas,and 0.85 atm ammonia gas is ________.
A) -1.0
B) -4.09 × 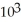
C) -8.62 × 103
D) -118.0
E) -41.9
Correct Answer:
Verified
Q99: The value of ΔG° at 181.0 °C
Q100: What is the equilibrium constant for a
Q101: At what temperature (in K)will a reaction
Q102: What is the equilibrium constant for a
Q103: A reversible change produces the maximum amount
Q105: For a given reaction with ΔS =
Q106: For a given reaction,ΔH = +74.6 kJ/mol,and
Q107: For the reaction C(s)+ Q108: For a given reaction,ΔS = +69.0 J/mol∙K,and Q109: At what temperature (in K)will a reaction![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents