The most useful ore of aluminum is bauxite,in which Al is present as hydrated oxides, 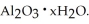 The number of kilowatt-hours of electricity required to produce
The number of kilowatt-hours of electricity required to produce 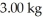 of aluminum from electrolysis of compounds from bauxite is ________ when the applied emf is
of aluminum from electrolysis of compounds from bauxite is ________ when the applied emf is 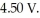
A) 0.0249
B) 0.0402
C) 26.8
D) 13.4
E) 40.2
Correct Answer:
Verified
Q86: How many grams of Cu are obtained
Q87: The electrolysis of molten Q88: How many grams of Ca metal are Q89: Based on standard reduction potentials,the most difficult Q90: In the formula ΔG = -nFE,n is Q92: The town of Natrium,West Virginia,derives its name Q93: The standard emf for the cell using Q94: How many minutes will it take to Q95: How many minutes will it take to Q96: The potential (E)to move K+ from the![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents