A rock contains 0.153 mg of lead-206 for each milligram of uranium-238.The 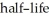 for the decay of uranium-238 to lead-206 is 4.5 × 109 yr.The rock was formed ________ years ago.
for the decay of uranium-238 to lead-206 is 4.5 × 109 yr.The rock was formed ________ years ago.
A) 6.89 × 108
B) 5.60 × 108
C) 7.33 × 108
D) 1.06 × 109
E) 8.08 × 108
Correct Answer:
Verified
Q124: The half-life of 218Po is 3.1 minutes.How
Q125: Cesium-131 has a half-life of 9.7 days.What
Q126: Q127: The beta decay of cesium-137 has a Q128: What percentage of a radioactive sample remains Q130: How much energy (in J)is produced when Q131: The decay of a radionuclide with a Q132: If we start with 1.000 g of Q133: If we start with 1.000 g of Q134: A freshly prepared sample of curium-243 undergoes![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents