A substance consisting of which molecule shown below should have a higher boiling point? 
A) The molecule on the left, CO 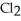 , because it has a fewer number of atoms.
, because it has a fewer number of atoms.
B) The molecule on the right, 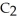
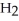 Cl, because it has a greater number of atoms.
Cl, because it has a greater number of atoms.
C) The molecule on the left, CO 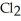 , because it is less symmetrical.
, because it is less symmetrical.
D) The molecule on the right, 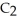
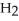 Cl, because of electronegativity considerations.
Cl, because of electronegativity considerations.
Correct Answer:
Verified
Q105: Which molecule is most polar?
A)S=C=S
B)O=C=O
C)O=C=S
D)These all have
Q141: Ammonia, N Q142: Which of the following molecules has the Q142: An individual carbon-oxygen bond is polar. Yet Q143: Water, Q146: Three kids sitting equally apart around a Q148: Which of the following molecules should have Q150: Would ammonia, N Q151: Which shows atoms in order of increasing Q151: Which of the following molecules is the Unlock this Answer For Free Now! View this answer and more for free by performing one of the following actions Scan the QR code to install the App and get 2 free unlocks Unlock quizzes for free by uploading documents![]()
![]()
![]()

