Given the following diagram, describe what happens electronically between these two molecules. 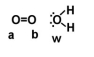
A) Oxygen B becomes slightly positively charged due to the electrons on the water molecule.
B) Oxygen A becomes slightly positively charged due to the electrons on the water molecule.
C) Oxygen W becomes slightly negatively charged due to the oxygen molecule.
D) Oxygen W becomes slightly positively charged due to the oxygen molecule.
E) none of the above
Correct Answer:
Verified
Q2: Fluorine is a relatively _.
A)large atom
B)soft atom
C)small
Q10: Which of the following is the weakest
Q15: Which of the following is the strongest
Q89: Which of the following substances is pure?
A)an
Q106: What is the difference between a dipole-dipole
Q109: If an ionic bond is stronger than
Q112: Which of the following molecules would you
Q115: Which of the following describes an aqueous
Q116: Which of the following molecules would you
Q122: What is a hydrogen bond?
A)a special type
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents