How are oxygen molecules attracted to water molecules?
A) The attraction between oxygen and water molecules is a classic example of dipole-dipole interaction.
B) The hydrogen bonding in water causes the attraction of the oxygen atoms in the 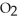 molecule to water.
molecule to water.
C) As a water molecule is brought close to an oxygen molecule an induced dipole results in the 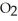 molecule causing the attraction.
molecule causing the attraction.
D) The attraction of oxygen and water molecules for one another is part of the common atom effect. Since both molecules contain oxygen, there is a built-in attraction.
Correct Answer:
Verified
Q27: A thin stream of water is pulled
Q32: List the following compounds in order of
Q35: Friends on a crowded ice skating rink
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents