Which solute graphed above has a solubility in water that does not markedly increase with increasing temperature?
A) NaN 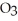
B) LiCl
C) KCl
D) NaCl
Correct Answer:
Verified
Q44: What is the molarity when water is
Q47: How many moles of water are there
Q49: If you need 10 moles of sucrose,how
Q61: Why does oxygen have such a low
Q61: Why does oxygen have such a low
Q64: Which of the following might have the
Q65: What property primarily determines the effect of
Q69: A sealed plastic bottle is filled with
Q73: What is the molarity of 0.50 liters
Q77: If the solubility of a compound is
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents