At 10°C, which is more concentrated-a saturated solution of sodium nitrate, NaN 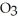 , or a saturated solution of sodium chloride? (See figure shown above )
, or a saturated solution of sodium chloride? (See figure shown above )
A) At 20°C a saturated solution of sodium nitrate, NaN 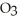 , is more concentrated than a saturated solution of sodium chloride, NaCl.
, is more concentrated than a saturated solution of sodium chloride, NaCl.
B) At 10°C a saturated solution of sodium chloride, NaCl, is more concentrated than a saturated solution of sodium nitrate, NaN 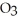 .
.
C) At 10°C a saturated solution of sodium nitrate, NaN 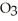 , is more concentrated than a saturated solution of sodium chloride, NaCl.
, is more concentrated than a saturated solution of sodium chloride, NaCl.
D) At 25°C a saturated solution of sodium nitrate, NaN 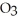 , is more concentrated than a saturated solution of sodium chloride, NaCl.
, is more concentrated than a saturated solution of sodium chloride, NaCl.
Correct Answer:
Verified
Q63: Describe what usually happens to a hot
Q68: Which of the following might have the
Q72: The air that a scuba diver breathes
Q74: If you were to increase the pressure
Q76: How is the solubility of a gas
Q78: How is the solubility of a solid
Q88: Based on atomic size, which would you
Q88: How can you tell whether a sugar
Q89: If nitrogen, Q89: If the solubility of a compound is![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents