The following diagram depicts the reaction between gaseous oxygen, 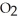 , and gaseous hydrogen,
, and gaseous hydrogen, 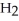 , to form water vapor,
, to form water vapor, 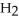 O. What molecules and how many of them should be drawn in the empty box? How many grams of water are formed under these conditions? How many grams of which chemical remain unreacted?
O. What molecules and how many of them should be drawn in the empty box? How many grams of water are formed under these conditions? How many grams of which chemical remain unreacted? 
A) five 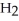 molecules in empty box; 9 grams of
molecules in empty box; 9 grams of 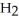 O + 0.5 grams of
O + 0.5 grams of 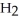
B) five 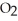 molecules in empty box; 9 grams of
molecules in empty box; 9 grams of 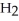 O + 8 grams of
O + 8 grams of 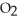
C) five 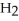 O molecules in empty box; 11.3 grams of
O molecules in empty box; 11.3 grams of 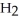 O + 5.7 grams of
O + 5.7 grams of 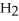 O
O
D) ten 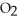 molecules in empty box; 9 grams of
molecules in empty box; 9 grams of 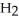 O + 8 grams of
O + 8 grams of 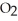
Correct Answer:
Verified
Q3: For the following balanced reaction, which of
Q12: For the following balanced equation,which has the
Q16: What is wrong with the following depiction
Q17: Which equations are balanced?
A. Mg (s)
Q20: What coefficients balance the following equation? _
Q26: Which is greater: 1.01 amu of hydrogen
Q30: If it takes 20 beryllium atoms to
Q31: The above diagram represents a chemical reaction
Q32: If it takes 200 golf balls to
Q40: Is it possible to have a macroscopic
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents