Small samples of oxygen gas needed in the laboratory can be generated by any number of simple chemical reactions, such as 2 KCl 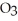 (s) → 2 KCl (s) + 3
(s) → 2 KCl (s) + 3 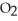 (g)
(g)
What mass of oxygen (in grams) is produced when 122.6 g of KCl 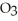 (formula mass = 122.6 amu) takes part in this reaction?
(formula mass = 122.6 amu) takes part in this reaction?
A) 32.00 grams
B) 48.00 grams
C) 96.00 grams
D) More information is needed.
Correct Answer:
Verified
Q43: What is the number of grams of
Q53: What is the mass of an oxygen
Q74: How many grams of gallium are there
Q75: Which has the greatest number of atoms?
A)
Q77: Two amu equals how many grams? (1
Q79: Which has the greatest number of molecules?
A)
Q80: There are 1000 liters in 1 cubic
Q110: Are the chemical reactions that take place
Q115: How many moles of H2 bonds are
Q120: ![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents