Given the following energy profiles, which of the following reactions is endothermic? 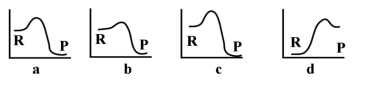 R= reactants P= products
R= reactants P= products
A) a
B) b
C) c
D) d
E) none of the above
Correct Answer:
Verified
Q68: Why might increasing the temperature alter the
Q82: In a chemical reaction, the bonds being
Q87: What is an exothermic reaction?
A)It is a
Q98: What is an endothermic reaction?
A)It is a
Q98: How many grams of water, 
Q103: Given that the bond energy of N2
Q105: How many bonds between nitrogen and hydrogen
Q109: If it takes energy to break bonds
Q113: Which of the following reaction energies is
Q114: Which of the following reaction energies is
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents