A fictional element has two naturally occurring isotopes with the natural abundances shown here: 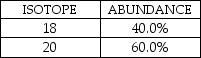
Which statement is TRUE for this element?
A) The atomic mass would be less than 18.
B) The atomic mass would be closer to 18 than to 20.
C) The atomic mass would be exactly 19.
D) The atomic mass would be closer to 20 than to 18.
E) The atomic mass would be greater than 20.
Correct Answer:
Verified
Q89: An isotope represented as S-34:
A)is an isotope
Q92: What is the mass number of the
Q96: There is very little of the element
Q97: The nucleus of an atom consists mainly
Q99: A specific isotope of an element is
Q100: A fictional element named Nivadium is found
Q101: What is the charge on a lithium
Q106: Given that the molecular mass of bromine
Q108: A fictional element has two isotopes and
Q111: A fictional element has two isotopes,each making
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents