An iron ore sample is found to be 35.00% Fe by mass.How many grams of ore are needed to obtain 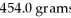
Of Fe?
A) 1297
B) 158.9
C) 295.1
D) 350.0
E) none of the above
Correct Answer:
Verified
Q66: One mole of potassium sulfate contains:
A)4 moles
Q69: If 2.01 × 1023 atoms of an
Q70: One mole of (NH4)2HPO4 contains how many
Q71: How many moles of carbon are in
Q72: How many moles of fluorine are in
Q73: If a sample of carbon dioxide contains
Q74: How many molecules of nitrogen monoxide are
Q79: Calculate the molar mass of ammonium carbonate.
A)78.05
Q83: What is the mass percent of chlorine
Q91: What is the mass percent of hydrogen
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents