What is the molecular formula of a compound given the molar mass of the compound is 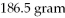
And the empirical formula is C2H7?
A) C4H14
B) C3H21
C) C2H7
D) C2H14
E) none of the above
Correct Answer:
Verified
Q82: Determine the correct empirical formula of a
Q83: What is the mass percent of chlorine
Q84: Which of the following compounds has the
Q85: An iron chloride compound contains 55.85 grams
Q86: Determine the empirical formula of a compound
Q88: Which of the following statements about empirical
Q90: The chemical formula CH2O can be classified
Q95: An estrogen compound with the empirical formula
Q97: A 7.96 gram sample of silver reacts
Q98: A chromium oxide compound contains 104.0 grams
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents