If you have 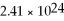
Atoms of copper,how many moles of copper do you have?
A) 0.250
B) 4.00
C) 0.600
D) 3.00
E) none of the above
Correct Answer:
Verified
Q83: What is the mass percent of chlorine
Q85: An iron chloride compound contains 55.85 grams
Q87: What is the mass percent of carbon
Q89: The empirical formula of a compound:
A)describes the
Q99: Determine the empirical formula of a compound
Q102: What is the mass of 
Q102: A compound has a molar mass of
Q103: Given that sodium chloride is 39.0% sodium
Q105: How many atoms of lithium are in
Q109: What is the mass percent of oxygen
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents