The ideal gas law is:
A) PV = nRT
B) P = 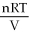
C) T = 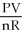
D) V = 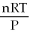
E) All of the above are forms of the ideal gas law.
Correct Answer:
Verified
Q79: What is the proper form of the
Q79: A gas sample occupies 3.50 liters of
Q80: Which one of the following is impossible
Q81: How many moles of gas were added
Q81: What is the major component of the
Q86: For an ideal gas,which of the following
Q87: Avogadro's Law is expressed as:
A)V is proportional
Q91: Which conditions can cause nonideal gas behavior
Q96: Which of the following gas law relationships
Q97: What problem could happen if deep sea
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents