A 250 gram sample of water at the boiling point had 45.0 kJ of heat added.How many grams of water were vaporized? Heat of vaporization for water is 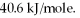
A) 1.11
B) 20.0
C) 0.902
D) 16.2
E) none of the above
Correct Answer:
Verified
Q96: Substance A is a molecular compound that
Q98: Which intermolecular forces are found in
Q99: Which compound in liquid form will have
Q102: How many kJ of heat are needed
Q103: What are the principal forces holding ice
Q107: How many kJ of heat are needed
Q108: Silicon is which type of solid?
A)molecular solid
B)ionic
Q108: How many kJ of heat are needed
Q109: Copper is which type of solid?
A)molecular solid
B)ionic
Q114: Which atomic solid has the highest melting
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents