A solution that is 35 percent by mass NaCl contains 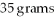
of NaCl dissolved in 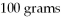
of water.
Correct Answer:
Verified
Q23: Colligative properties are independent of the amount
Q25: A stock solution is a more concentrated
Q27: A 0.10 molality solution of the sugar
Q30: The molarity of a solution prepared by
Q31: The solubility of gases in water increases
Q32: One liter of a 2.0 M NaOH
Q34: Molarity is defined as the moles of
Q35: A sample of salt water will freeze
Q36: Molality is calculated by dividing grams of
Q40: The terms "molarity"and "molality" are considered identical
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents