What is the molarity of a solution prepared by dissolving 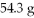
Of Ca(N 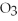
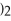
Into 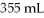
Of water?
A) 0.331 M
B) 0.932 M
C) 0.117 M
D) 1.99 M
E) none of the above
Correct Answer:
Verified
Q63: How many grams of LiF would be
Q72: What is the mass percent of an
Q72: We dissolve 2.45 g of sugar in
Q73: What is the mass percent of a
Q74: What is the molarity of a solution
Q74: The solubility of gases in water:
A)is independent
Q76: How many grams of KCl are needed
Q79: When preparing sodium hydroxide solution,it is best
Q88: What mass (in grams)of fructose (C6H12O6)is contained
Q100: What volume of 12.0 M HCl is
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents