A 25.0 mL sample of 0.105 M HCl was titrated with 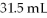
Of NaOH.What is the concentration of the NaOH?
A) 0.0833 M
B) 0.132 M
C) 0.105 M
D) 0.075 M
E) none of the above
Correct Answer:
Verified
Q62: In the following reaction: HCO3- (aq)+ H2O
Q63: Which of the following acids is a
Q64: When an acid reacts with a metal,what
Q65: What are the products of a neutralization
Q66: A neutralization reaction between KOH (aq)and H2SO4
Q71: Which of the following acids is diprotic?
A)HCl
Q73: A substance that acts as an acid
Q74: A 35.0 mL sample of 0.225 M
Q76: Which of the following is a weak
Q78: Which of the following is NOT an
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents