The titration of 25.00 mL a 0.125 M HCl 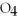
Solution required 21.37 mL of KOH to reach the endpoint.What is the concentration of the KOH?
A) 0.292 M
B) 0.0668 M
C) 0.146 M
D) 0.134 M
E) none of the above
Correct Answer:
Verified
Q102: Which of the following contributes to acid
Q104: A buffer solution is all of the
Q106: Which of the following statements are TRUE
Q107: What is the pH of a solution
Q109: Which combination below will be a buffer
Q111: Acid rain legislation targeted the release of
Q112: What is the [H⁺] in a solution
Q113: What is the pH of a solution
Q114: Which solution below would be considered the
Q115: What is the concentration of 
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents