Multiple Choice
What is the oxidation state of the underlined atom in the reaction: 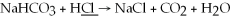
A) 0
B) +1
C) -1
D) +2
E) -2
Correct Answer:
Verified
Related Questions
Q43: A sacrificial electrode works by being reduced
Q46: Which of the following are typically TRUE
Q51: The oxidation number of Cr in Cr2O7
Q59: Identify the substance being reduced in the
Q66: What is the oxidation state of the
Q67: For the reaction KMnO4 + Li →
Q68: The oxidation state of Na in Na2SO4
Q73: For the reaction Co + Cl2 →
Q76: Assign the oxidation state of each atom
Q77: What is the oxidation state of nitrogen
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents