Magnesium (used in the manufacture of light alloys) reacts with iron(III) chloride to form magnesium chloride and iron. 3Mg(s) + 2FeCl3(s) 3MgCl2(s) + 2Fe(s)
A mixture of 41.0 g of magnesium ( 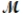 = 24.31 g/mol) and 175 g of iron(III) chloride (
= 24.31 g/mol) and 175 g of iron(III) chloride ( 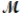 = 162.2 g/mol) is allowed to react. Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.
= 162.2 g/mol) is allowed to react. Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.
A) Limiting reactant is Mg; 67 g of FeCl3 remain.
B) Limiting reactant is Mg; 134 g of FeCl3 remain.
C) Limiting reactant is Mg; 104 g of FeCl3 remain.
D) Limiting reactant is FeCl3; 2 g of Mg remain.
E) Limiting reactant is FeCl3; 87 g of Mg remain.
Correct Answer:
Verified
Q60: An important reaction sequence in the
Q61: Analysis of a white solid produced in
Q62: One mole of O2 has a mass
Q63: What is the percent yield for
Q64: For a sample consisting of 2.50 g
Q66: Ammonia, NH3, is produced industrially from
Q67: Gaseous methanol (CH4O) reacts with oxygen gas
Q68: Balance the equation
B2O3(s) + NaOH(aq)
Q69: In 0.20 mole of phosphoric acid, H3PO4
a.
Q70: Methanol (CH4O) is converted to bromomethane
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents