Diborane (B2H6) has been considered as a possible rocket fuel. Calculate H° for the reaction
B2H6(g) 2B(s) + 3H2(g)
using the following data: 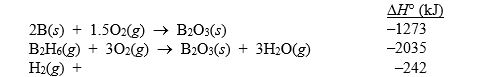
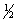 O2(g) H2O(g)
O2(g) H2O(g)
-242
Correct Answer:
Verified
Q7: For all processes, both q and w
Q13: The enthalpy (H) of liquid water is
Q14: The only way in which a system
Q61: For a reaction in a sealed,
Q64: Clearly state the thermodynamic standard state of
a.
Q65: Although internal energy (E) is more
Q68: The reaction
2NaOH(aq) + H2SO4(aq)
Q69: A mass of 1.250 g of benzoic
Q70: a. Starting from the equation H
Q71: a. State Hess's Law.
b. Use the
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents