Refer to the table below. 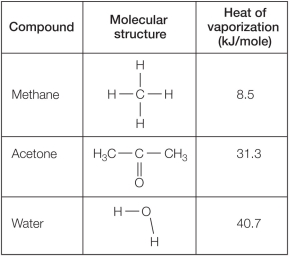 Which statement explains reasons for differences in the heat of vaporization for these compounds?
Which statement explains reasons for differences in the heat of vaporization for these compounds?
A) Methane has the lowest heat of vaporization because its molecules experience no intermolecular forces of attraction.
B) Water has the highest heat of vaporization because it is the smallest in mass and physical size.
C) Both acetone and water have higher heat of vaporization compared with methane because both are polar molecules capable of hydrogen bonding.
D) Water has a higher heat of vaporization compared with acetone and methane because it is a polar molecule and the other two are not.
E) Water has the highest heat of vaporization because it is the only compound that is both polar and capable of forming hydrogen bonds.
Correct Answer:
Verified
Q190: Consider two scenarios: (1) a living cell
Q191: Refer to the figures below. 
Q192: Which statement best explains how atoms change
Q193: The Australian environmental protection agency defines bioremediation
Q194: When a chemical reaction occurs, changes in
Q196: Which compound is expected to have a
Q197: Consider the following statement: In a chemical
Q198: Although air temperatures at a site on
Q199: Which general chemical principles can be applied
Q200: Refer to the figures below. 
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents