Refer to the table below. 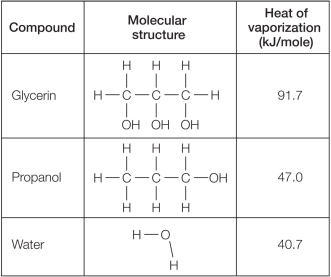 Which statement explains reasons for differences in the heat of vaporization for these compounds?
Which statement explains reasons for differences in the heat of vaporization for these compounds?
A) Glycerin's high heat of vaporization compared with that of the other molecules can be explained because it is both polar and capable of forming hydrogen bonds.
B) The heat of vaporization of glycerin is twice as high as that of water because glycerin is so much larger in size than water.
C) The greater hydrogen-bonding capacity of glycerin explains its greater heat of vaporization when compared with water or propanol.
D) Water has a lower heat of vaporization compared with glycerin and propanol because its hydrogen bonds are weaker.
E) Molecules that are polar and capable of forming hydrogen bonds tend to have similar heats of vaporization.
Correct Answer:
Verified
Q178: Which statement about spiders illustrates the importance
Q179: How does the ionic compound magnesium chloride
Q180: Five statements describing water are given below.Which
Q181: Refer to the table and three diagrams
Q182: In an organic chemistry lab, chemical reactions
Q184: Refer to the figure below. 
Q185: The equation C4H10 + 7 O2
Q186: Refer to the balanced chemical equation
Q187: Which set of numbers correctly balances
Q188: Refer to the balanced chemical equation
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents