System 1 is 4.30 moles of a monatomic ideal gas held at a constant volume as it undergoes a temperature increase.System 2 is 4.30 moles of a diatomic ideal gas held at constant volume as it also undergoes a temperature increase.The thermal energy absorbed by System 1 is 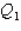 ,and the thermal energy absorbed by System 2 is
,and the thermal energy absorbed by System 2 is 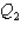 as both systems undergo a temperature increase of 75.0 K.What is the ratio
as both systems undergo a temperature increase of 75.0 K.What is the ratio 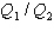 ?
?
A) 1.00
B) 0.600
C) 1.67
D) The volumes of the systems need to be known before the ratio can be found.
Correct Answer:
Verified
Q5: Which of the following increases the internal
Q6: An adiabatic expansion refers to the fact
Q16: Area on a PV diagram has units
Q17: A 2.0-mol ideal gas system is maintained
Q18: A system is acted on by its
Q19: The adiabatic index of a gas is
Q22: A heat engine exhausts 3 000 J
Q23: A heat engine receives 6 000 J
Q25: A turbine takes in 1 000-K steam
Q29: A thermodynamic process that happens very quickly
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents