A 3.31-g sample of lead nitrate, 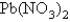 ,molar mass = 331 g/mol,is heated in an evacuated cylinder with a volume of 2.37 L.The salt decomposes when heated,according to the equation:
,molar mass = 331 g/mol,is heated in an evacuated cylinder with a volume of 2.37 L.The salt decomposes when heated,according to the equation: 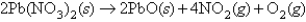 Assuming complete decomposition,what is the pressure in the cylinder after decomposition and cooling to a temperature of 300.K? Assume the
Assuming complete decomposition,what is the pressure in the cylinder after decomposition and cooling to a temperature of 300.K? Assume the 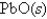 takes up negligible volume.
takes up negligible volume.
A) 0.260 atm
B) 0.208 atm
C) 0.0519 atm
D) 0.364 atm
E) 34.4 atm
Correct Answer:
Verified
Q42: Into a 2.22-liter container at 25°C are
Q64: The purity of a sample containing zinc
Q65: A 1.00-g sample of a gaseous compound
Q67: Standard pressure for gases is
A)0 atm
B)1 atm
C)100
Q68: One way to isolate metals from their
Q70: What volume of carbon dioxide measured at
Q71: If M is the molar mass,R the
Q72: The standard temperature for gases is
A)100°C
B)0°C
C)32°C
D)212°F
E)0°F
Q73: What volume does 40.5 g of N2
Q74: What volume of ![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents