Solved
The Decomposition of N2O5(g)to NO2(g)and O2(g)obeys First-Order Kinetics 10-5 S-1 at 25°C,what Is the Half-Life for the Reaction
Multiple Choice
The decomposition of N2O5(g) to NO2(g) and O2(g) obeys first-order kinetics.Assuming the form of the rate law is: 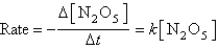
Where k = 5.4 10-5 s-1 at 25°C,what is the half-life for the reaction described?
A) 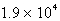 s
s
B) 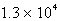 s
s
C) 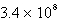 s
s
D) 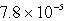 s
s
E) none of these
Correct Answer:
Verified
Related Questions
Q72: The elementary chemical reaction O +
Q73: Two isomers (A and B)of a
Q74: Consider the reaction 2O3(g)
Q75: The decomposition of ozone may occur through
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents