A chemist is planning to study a chemical equilibrium for which no measured explicit thermodynamic values are available. However, the reaction involves common chemical species. What information could a chemist use to make predictions about the equilibrium?
A) equilibrium constants for the equilibrium reactions that can be combined to make the new equilibrium
B) (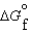 values for the reactants and products of the new reaction)
values for the reactants and products of the new reaction)
C) ( Hf and S values for the reactants and products of the new reaction)
D) all of the above
Correct Answer:
Verified
Q145: An oxygen molecule can have several different
Q146: Of the three modes of molecular motion-vibration,
Q147: The entropy of an oxygen molecule
Q148: What is the overall standard free-energy
Q149: The standard entropy of nitrogen gas
Q151: Boltzmann derived the relationship S = k
Q152: Which statement is true of the ideal
Q153: What type of motion is depicted in
Q154: Noxious NO gas can form from
Q155: Which of the following states of motion
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents