Phosphoric acid is a triprotic acid, ionizing in the following sequential steps: H3PO4 + H2O  H2PO4- + H3O+
H2PO4- + H3O+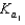 H2PO4- + H2O
H2PO4- + H2O  HPO42- + H3O+
HPO42- + H3O+ 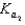
HPO42- + H2O  PO43- +H3O+
PO43- +H3O+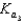
Which equilibrium is most important in determining the pH of a solution of sodium phosphate?
A) HPO42- + H2O  PO43- + H3O+
PO43- + H3O+
B) H3PO4 + H2O  H2PO4-+ H3O+
H2PO4-+ H3O+
C) PO43-+ H2O  HPO42-+ OH-
HPO42-+ OH-
D) H2PO4- + H2O  H3PO4 + OH-
H3PO4 + OH-
E) 2H2O  H3O++ OH-
H3O++ OH-
Correct Answer:
Verified
Q112: The concentration of acetic acid in vinegar
Q113: The stronger the acid, _
A)the stronger its
Q114: What is the pH of a
Q115: A cleaning solution has a pOH of
Q116: Define a conjugate acid-base pair and provide
Q117: Explain why some salts produce neutral solutions
Q119: Methylamine (CH3NH2) is a weakly basic compound.
Q120: You decide to put your growing knowledge
Q121: What is the pH of a
Q122: What is the pH of a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents