Oxygen for astronauts in space can be produced chemically using a solid fuel oxygen generator. The reaction takes place at 600 C and is shown below. NaClO3(s) 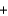 Fe(s)
Fe(s)  3O2(g)
3O2(g) 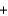 NaCl(s)
NaCl(s) 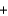 FeO(s)
FeO(s)
What volume of O2 gas will be produced when 855 g of NaClO3 reacts with excess iron at a pressure of 3 atm?
A) 575 L
B) 2.80 L
C) 192 L
D) 6.13 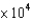 L
L
E) 396 L
Correct Answer:
Verified
Q70: The density of a pure gas at
Q71: Indicate the gas with the greatest density
Q72: What is the pressure of NO
Q73: Which of the following samples of an
Q74: Which one of the following gases could
Q76: Poisonous carbon monoxide can be produced by
Q77: Which of the following graphs shows how
Q78: Which of the following gases has nearly
Q79: The density of a gaseous compound of
Q80: The density of an unknown gas
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents