Determine the root-mean-square speed of methane (CH4) molecules that have an average kinetic energy of 10.5 J/mol.
A) 337 m/s
B) 1.14 m/s
C) 2.81 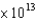 m/s
m/s
D) 36.2 m/s
E) 1,150 m/s
Correct Answer:
Verified
Q98: Which container shown below has the largest
Q99: Which container shown below has the largest
Q100: When a certain acid and a certain
Q101: The root-mean-square speed of a gas is
Q102: Hydrogen gas is being promoted as
Q104: Helium-neon (HeNe) lasers contain a low-pressure mixture
Q105: Which one of the following statements is
Q106: Which of these gases (He, N2O,
Q107: Which one of the following statements is
Q108: What is the root-mean-square speed of N2O
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents