Which one of the following statements about the properties of gases is not correct?
A) At low temperatures, the attractive forces between molecules cause a real gas to deviate from ideal gas behavior.
B) At high pressures, the finite size of the molecules causes a real gas to deviate from ideal gas behavior.
C) The ratio PV/RT = 1 for one mole of an ideal gas.
D) The ratio PV/nRT 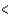 1 because of repulsive interactions between the molecules.
1 because of repulsive interactions between the molecules.
E) The ratio PV/nRT 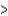 1 because of the finite size of the molecules.
1 because of the finite size of the molecules.
Correct Answer:
Verified
Q128: What pressure (in Pa) will be exerted
Q129: The diffusion rate of H2 in a
Q130: How high (in feet) can one atmosphere
Q131: Which of the following gases will effuse
Q132: Which set of gases is listed from
Q134: _ is how gases spread among each
Q135: Which statement A-D below is not true
Q136: Which set of gases is listed from
Q137: The root-mean-square speeds of five gases at
Q138: A gas was found to diffuse twice
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents