Oxygen for astronauts in space can be produced chemically using a solid fuel oxygen generator. The reaction takes place at 600 C and is shown below.
NaClO3(s) 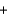 Fe(s)
Fe(s)  3O2(g)
3O2(g) 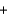 NaCl(s)
NaCl(s) 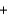 FeO(s)
FeO(s)
What volume of O2 gas will be produced when 855 g of NaClO3 reacts with excess iron at a pressure of 3 atm?
Correct Answer:
Verified
Q144: Before the development of reliable batteries, miners'
Q145: Hyperbaric oxygen chambers providing pure oxygen (O2)
Q146: Which one of these samples contains
Q147: A scuba diver releases a balloon containing
Q148: Three bulbs are connected as shown in
Q150: In terms of the kinetic molecular theory,
Q151: A 5 L diving tank is filled
Q152: A sample of gas at 30
Q153: He and O2 gases are mixed at
Q154: Plot the relationship between each of the
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents