You hold a 50 g sphere of copper in one hand and a 25 g sphere of aluminum in the other hand. If both absorb energy at the same rate, which will come to your body temperature first and why? The specific heat capacities are 0.4 J/(g . 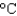 ) for copper and 0.9 J/(g .
) for copper and 0.9 J/(g . 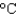 ) for aluminum.
) for aluminum.
A) copper, because the specific heat is smaller
B) aluminum, because the specific heat is larger
C) aluminum, because the mass is smaller
D) copper, because the heat capacity is smaller
E) Both reach body temperature at the same time because they absorb energy at the same rate.
Correct Answer:
Verified
Q70: The cooling system in an automobile holds
Q71: In an experiment, 5.0 g of ice
Q72: A 150 g piece of iron (CP
Q73: In an experiment, 10.0 g of
Q74: The energy content of a Big Mac
Q76: A food sample was burned in
Q77: You have a summer job in a
Q78: In an experiment, 74.3 g of
Q79: Using the following data for water, determine
Q80: In an experiment, 7.5 g of
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents