Silver salts are used in black-and-white photography. Owing to the value of silver, the ions left in developing solutions are often collected by precipitation reactions for recycling. A 4.5 L jug of a developing solution was treated with an excess of sodium chloride, and a precipitate containing 2.86 g of dry silver chloride was collected. What was the molar concentration of the silver ions in the developing solution? The net ionic equation is
Ag+(aq) 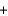 Cl-(aq)
Cl-(aq)  AgCl(s)
AgCl(s)
A) 2.9 M
B) 5.9 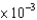 M
M
C) 2.9 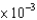 M
M
D) 4.4  M
M
E) 4.4 M
Correct Answer:
Verified
Q87: The oxidation number of carbon in carbon
Q88: The thermite reaction is used in welding
Q89: Which one of the following statements is
Q90: Write the complete ionic equation for the
Q91: Ion-exchange resins can remove Mg2+ and Ca2+0
Q93: What mass of lead(II) chloride is produced
Q94: What mass of barium sulfate (molar mass
Q95: What mass of silver chloride will be
Q96: A reaction that takes place between dissolved
Q97: What is the oxidation number of S
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents