Which of the following graphs shows the correct relationship between the pressure and temperature of an ideal gas in a given volume? Note the origin corresponds to P = 0 and T = 0.
A) 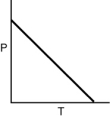
B) 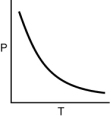
C) 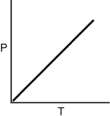
D) 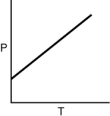
Correct Answer:
Verified
Q1: What pressure (in Pa) will be
Q3: What force is exerted by exactly
Q4: The atmospheric pressure is reported to be
Q6: If the pressure in the eye of
Q7: What pressure will be measured (in
Q11: Which of the following substances will require
Q15: What height of water (in meters) is
Q16: The pressure of a gas is inversely
Q20: A barometer measures a pressure of 745
Q40: What is the volume of a gas
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents