Which of the following reactions will result in a reduced total pressure?
A) CH4(g) + 2O2(g) CO2(g) + 2H2O(g)
B) 2N2O(g) 2N2(g) + O2(g)
C) 2HI(g) H2(g) + I2(g)
D) 2H2(g) + O2(g) 2H2O( 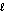 )
)
Correct Answer:
Verified
Q24: Which of the gases shown here will
Q24: What volume is occupied by 1.00 mol
Q26: A 1.50 mol sample of He occupies
Q31: How many moles of propane are contained
Q32: An unknown gas held in a 25.0
Q33: In an experiment, 7.5 mol of a
Q35: What is the pressure of a 3.5
Q54: The pressure gauge on a cylinder used
Q59: What is the temperature if 5.0 g
Q70: The density of a pure gas at
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents