Calculate G° for 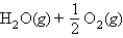

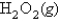 at 600. K, using the following data: H2(g) + O2(g)
at 600. K, using the following data: H2(g) + O2(g)  H2O2 Kp = 2.3 * 106 at 600. K
H2O2 Kp = 2.3 * 106 at 600. K
2H2(g) + O2(g)  2H2O(g) Kp = 1.8 * 1037 at 600. K
2H2O(g) Kp = 1.8 * 1037 at 600. K
A) -220 kJ
B) -350 kJ
C) +140 kJ
D) -290 kJ
E) +290 kJ
Correct Answer:
Verified
Q97: In which reaction is
Q98: Consider the gas phase reaction
Q99: Assume that the reaction 
Q100: Consider the gas phase reaction
Q101: Given the following free energies of formation:
Q102: What will be the effect on the
Q104: Consider a weak acid, HX. The
Q105: Consider the reaction 2SO2(g) + O2(g)
Q107: The standard free energy of formation of
Q110: Consider the following system at equilibrium at
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents