Copper has two naturally occurring isotopes with atomic masses of 62.9296 u ( 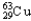 ) and 64.9278 u (
) and 64.9278 u ( 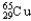 ) .The atomic mass of copper is 63.546 u.What is the percent distribution of the isotopes?
) .The atomic mass of copper is 63.546 u.What is the percent distribution of the isotopes?
A) 69.15% 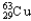 ,30.85%
,30.85% 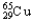
B) 30.85% 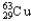 ,69.15%
,69.15% 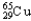
C) 1.08% 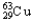 ,98.92%
,98.92% 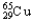
D) 98.92% 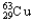 ,1.08%
,1.08% 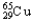
E) 99.03% 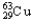 ,0.97%
,0.97% 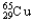
Correct Answer:
Verified
Q21: The natural distribution of the isotopes of
Q24: Groups in the periodic table are also
Q25: Which of the following is the best
Q34: One of the main features of Dalton's
Q35: Which of the following gives the correct
Q40: Which of the following is correct for
Q44: Consider the following figure. Q45: Which of the following is the correct Q47: Which of the following symbol/name pairs is/are Q48: Which of the following is the correct![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents