Consider the following representation of a gaseous chemical reaction. 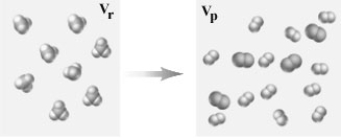 If carried out at constant temperature and pressure how would the volume in the container before the reaction (Vr) compare to that after the reaction (Vp) ?
If carried out at constant temperature and pressure how would the volume in the container before the reaction (Vr) compare to that after the reaction (Vp) ?
A) Vr = Vp
B) Vr = 2Vp
C) 2Vr = Vp
D) 3Vr = Vp
E) Vr = 3Vp
Correct Answer:
Verified
Q5: Consider the algebraic expression a* b
Q6: The STP density of a gas is
Q7: The density of an unknown gas is
Q8: Calculate the number of moles in 3.13
Q9: Which of the following is a mathematical
Q11: Consider the balloon shown below which has
Q12: Determine the density of nitrogen gas at
Q13: A gas has a density of 1.54
Q15: What volume is occupied by 0.0100 mole
Q20: What is the common reference conditions for
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents