Suppose white atoms (W) and black atoms (B) react according to the following balanced chemical equation. 2W + B → BW2
If the initial reaction mixture is prepared as follows,what will the final reaction mixture be once the reaction is complete? 
A) 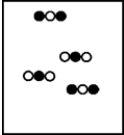
B) 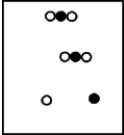
C) 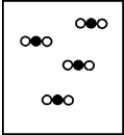
D) 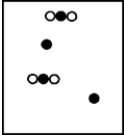
E) 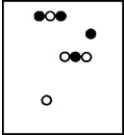
Correct Answer:
Verified
Q68: In a correctly balanced equation, the number
Q101: What is the theoretical yield of chromium
Q112: What is the theoretical yield of aluminum
Q113: Which is a representation of a balanced
Q113: There is only one distinct empirical formula
Q116: Which could represent the reaction mixture after
Q116: One way of obtaining pure sodium carbonate
Q120: The empirical formula of C6H6 is CH.
Q121: The _ is the amount of product
Q134: The percent yield can be determined by
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents