What is the mass density of argon gas at pressure 1.00 × 105 N/m2 and at temperature 300 K? The mean atomic mass of argon is 39.948 g/mol and the ideal gas constant is R = 8.314 J/mol ∙ K = 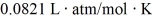 .
.
A) 1.40 kg/m3
B) 1.00 kg/m3
C) 1.20 kg/m3
D) 1.60 kg/m3
E) 1.80 kg/m3
Correct Answer:
Verified
Q33: A vertical tube that is closed at
Q34: 3.00 moles of an ideal gas at
Q35: How many moles of water (H2O)molecules are
Q35: A certain metal has a coefficient of
Q36: Sometimes an experiment requires a certain pure
Q38: A 3.2-L volume of neon gas (Ne)is
Q39: A cold trap is set up to
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents