Benzaldehyde ( 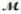 = 106.1 g/mol) , also known as oil of almonds, is used in the manufacture of dyes and perfumes and in flavorings. What would be the freezing point of a solution prepared by dissolving 75.00 g of benzaldehyde in 850.0 g of ethanol? Kf = 1.99°C/m, freezing point of pure ethanol = -117.3°C.
= 106.1 g/mol) , also known as oil of almonds, is used in the manufacture of dyes and perfumes and in flavorings. What would be the freezing point of a solution prepared by dissolving 75.00 g of benzaldehyde in 850.0 g of ethanol? Kf = 1.99°C/m, freezing point of pure ethanol = -117.3°C.
A) -117.5°C
B) -118.7°C
C) -119.0°C
D) -120.6°C
E) < -121°C
Correct Answer:
Verified
Q64: Calculate the freezing point of a solution
Q65: Hexachlorophene is used as a disinfectant in
Q67: Which one of the following pairs of
Q72: Safrole is used as a topical antiseptic.
Q75: Determine the freezing point of a solution
Q84: Lysine is an amino acid that is
Q88: An emulsion is a dispersion consisting of
Q92: The Tyndall effect
A)is observed in concentrated solutions.
B)is
Q95: Human blood has a molar concentration of
Q96: A 0.100 m K2SO4 solution has a
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents