Nitrosyl bromide decomposes according to the chemical equation below. 2 NOBr(g) 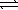 2 NO(g) + Br2(g)
2 NO(g) + Br2(g)
When 0.260 atm of NOBr is sealed in a flask and allowed to reach equilibrium,22% of the NOBr decomposes.What is the equilibrium constant,Kp,for the reaction?
A) 2.3 × 10-3
B) 4.5 × 10-3
C) 3.5 × 10-2
D) 4.8 × 10-2
E) 8.0 × 10-2
Correct Answer:
Verified
Q42: At a given temperature,K = 0.024 for
Q43: Given the following chemical equilibria, N2(g)+ O2(g)
Q44: Nitrogen and oxygen gases may react to
Q45: Sulfuryl chloride decomposes to sulfur dioxide and
Q46: For the equilibrium N2O4(g) Q48: In an experiment,0.46 mol H2 and 0.46 Q49: For the reaction N2O4(g) Q50: A 3.50-mol sample of HI is placed Q51: Consider the following equilibrium: CO2(g)+ H2(g) Q52: For the equilibrium PCl5(g) Unlock this Answer For Free Now! View this answer and more for free by performing one of the following actions Scan the QR code to install the App and get 2 free unlocks Unlock quizzes for free by uploading documents![]()
![]()
![]()

