For the equilibrium N2O4(g) 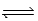 2NO2(g) ,at 298 K,Kp = 0.15.For this reaction system,it is found that the partial pressure of N2O4 is 3.7 × 10-2 atm at equilibrium.What is the partial pressure of NO2 at equilibrium?
2NO2(g) ,at 298 K,Kp = 0.15.For this reaction system,it is found that the partial pressure of N2O4 is 3.7 × 10-2 atm at equilibrium.What is the partial pressure of NO2 at equilibrium?
A) 4.9 atm
B) 24 atm
C) 0.0016 atm
D) 0.0055 atm
E) 0.074 atm
Correct Answer:
Verified
Q41: The equilibrium constant (Kc)for the decomposition of
Q42: At a given temperature,K = 0.024 for
Q43: Given the following chemical equilibria, N2(g)+ O2(g)
Q44: Nitrogen and oxygen gases may react to
Q45: Sulfuryl chloride decomposes to sulfur dioxide and
Q47: Nitrosyl bromide decomposes according to the chemical
Q48: In an experiment,0.46 mol H2 and 0.46
Q49: For the reaction N2O4(g) Q50: A 3.50-mol sample of HI is placed Q51: Consider the following equilibrium: CO2(g)+ H2(g) ![]()
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents