A solution is prepared by adding 1.40 g of solid NaCl to 50.0 mL of 0.100 mol L-1 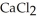 . What is the molarity of chloride ion in the final solution? Assume that the volume of the final solution is 50.0 mL.
. What is the molarity of chloride ion in the final solution? Assume that the volume of the final solution is 50.0 mL.
A) 0.228 mol L-1
B) 0.579 mol L-1
C) 0.479 mol L-1
D) 0.679 mol L-1
E) 0.128 mol L-1
Correct Answer:
Verified
Q7: What are the coefficients in front of
Q111: When dissolved in water,KOH behaves as:
A)an acid
Q156: What is the stoichiometric coefficient for oxygen
Q157: Which of the following compounds is an
Q158: Silver ions can be precipitated from aqueous
Q159: H Br , H I , H
Q160: Which of the following solutions will have
Q162: Lithium and nitrogen react to produce lithium
Q165: What is the oxidation number change for
Q166: Automotive air bags inflate when sodium azide
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents